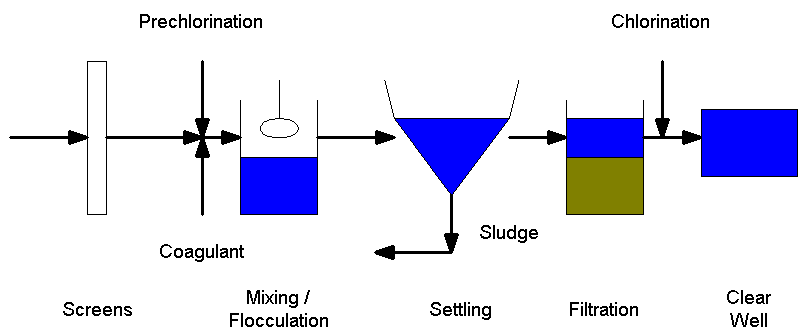
Pre-Chlorination & pH Correction
Pre-Chlorination
This is essential as it is used for disinfecting the water.Chlorine is added to get rid of bacteria micro germs.Without this crucial step, industrial water would not qualify as usable as it will be filled with substances that may cause unwanted side effects.
What is chlorine ?
Chlorine is obtained from salt (NaCl). It exists as a gas at atmospheric pressures but liquifies under pressure.
Chlorine is a highly efficient disinfectant, and is added to public water supplies to kill disease-causing pathogens. They include bacteria, viruses and protozoans. They commonly grow in water supply reservoirs, on the walls of water mains and in storage tanks. These microscopic agents of disease were the cause of many people’s death before the use of chlorine to disinfect.
Chlorine is a highly efficient disinfectant, and is added to public water supplies to kill disease-causing pathogens. They include bacteria, viruses and protozoans. They commonly grow in water supply reservoirs, on the walls of water mains and in storage tanks. These microscopic agents of disease were the cause of many people’s death before the use of chlorine to disinfect.
How exactly does chlorine kill bacteria and agents of diseases
As a strong oxidizing agent, chlorine kills via the oxidation of organic molecules.Chlorine and hydrolysis product hypochlorous acid are neutrally charged and therefore easily penetrate the negatively charged surface of pathogens. It is able to disintegrate the lipids that compose the cell wall and react with intracellular enzymesand proteins, making them nonfunctional. Micro organisms then either die or are no longer able to multiply.
pH Correction
This goes hand in hand with pre-chlorination.It is a procedure where caustic soda is added to maintain the desired pH level. This is to ensure that the water is not too corrosive. If the water is too acidic, unwanted side effects might happen. The first thing that would happen would be downtime for the factory and the repair cost might be extremely expensive.The quality of water they want to achieve would undoubtedly be 'pure' water, where the pH level is 7
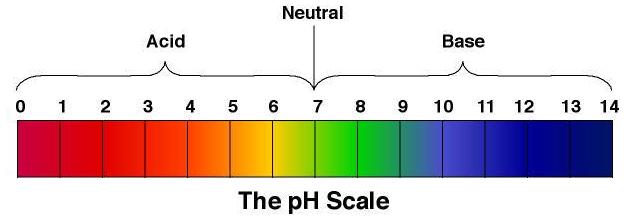
What exactly is pH level?
It is a measure of the acidity of an aqueous solution. It ranges from pH 1 to 14.
pH levels from 1 to 6 are acidic while pH levels from 8 to 14 are of alkaline nature.
a pH level of 7 would mean that it is neutral.
-------------------------------------------------------------------------------------------------------------------
Basically
1 to 6 = acids
7=neutral
8 to 14=alkaline nature/bases
pH levels from 1 to 6 are acidic while pH levels from 8 to 14 are of alkaline nature.
a pH level of 7 would mean that it is neutral.
-------------------------------------------------------------------------------------------------------------------
Basically
1 to 6 = acids
7=neutral
8 to 14=alkaline nature/bases